Molecular Diagnostics Nucleic Acid Isolation

Molecular Diagnostics Nucleic Acid Isolation
The Molecular Diagnostics section of the BMP Core Shared Resource offers nucleic acid isolation from a variety of different specimen types including blood, fresh or frozen tissue, and FFPE (including micro-dissection). Included in the price of each extraction is nucleic acid quantification by NanoDrop andgeneration of a nucleic acid isolation report. Additional fees apply for Biorepository and Research Histology Services and quantification by Qubit and Agilent TapeStation RIN analysis.
Human Specimens from the Biorepository under the General BioBanking and Using IRB Protocol
If requesting human specimens from the Biorepository through the general biobanking and using IRB protocol, please make a request to BMP-Biorepository@hci.utah.edu. The Biorepository staff will coordinate consent validation, specimen collection or retrieval, histology, and neoplastic cell content review by a pathologist and will deliver the specimens to the Molecular Diagnostics Laboratory for extraction. A GNomEx request for the nucleic acid isolation will be made on behalf of the investigator, and the sample will be de-identified and assigned a CC# through itBioPath. Upon completion, the investigator will be notified, and the nucleic acid isolation report will be available through GNomEx.
De-identified (exempt) Human and Animal Specimens
Please submit a GNomEx request directly to the Molecular Diagnostics Laboratory for de-identified (exempt) human and animal specimens. Please see the sample submission page for additional instructions.
Isolation Services
- DNA from blood, buffy coat, plasma or serum (Qiagen QIAamp DNA Blood Mini Kit Cat# 51185)
- Input: Up to 0.2 mL of fresh and frozen whole blood with common anticoagulants, such as EDTA, citrate, or heparin and buffy coat, PBMC, plasma or serum.
- Extraction Notes: Elution in 100 µL of buffer AE (10 mM Tris-HCl, 0.5 mM EDTA pH 9.0)
- DNA and RNA from fresh or frozen tissue (Qiagen AllPrep DNA/RNA/miRNA Universal Kit Cat# 80224)
- Input: 30 mg of fresh or frozen tissue
- Extraction Notes: RNA is eluted in 60 µL RNase-free water. and DNA is eluted in 100 µL of buffer EB (10 mM Tris-HCl pH 8.5).
- DNA FFPE (Qiagen QiaAMP DNA FFPE Kit Cat# 56404)
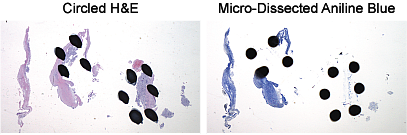
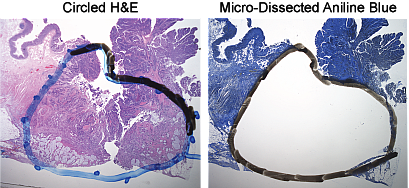
- Micro-Dissection Input: Circled H&E and 8 x 10 µm aniline blue sections with tissue area of 250 mm2 or 15-20 sections for smaller specimens and biopsies
- Scrolls and Punches Input: Maximum of 35 mg of material. Punches larger than 2-3 mm are challenging to digest.
- Extraction Details: RNase digestion prior to elution in 35 µL of buffer ATE (10 mM Tris-HCl, 0.1 mM EDTA, 0.04% Sodium Azide pH 8.3)
- RNA FFPE (Qiagen miRNeasy FFPE Kit Cat# 217504 )
- Micro-Dissection Input: Circled H&E and 8 x 10 µm aniline blue sections with tissue area of 250 mm2 or 15-20 sections for smaller specimens and biopsies
- Scrolls and Punches Input: Maximum of 35 mg of material. Punches larger than 2-3 mm are challenging to digest.
- Extraction Notes: On-column DNase digestion performed prior to elution in 20 µL RNase-free water.
FFPE Micro-Dissection
The Molecular Diagnostics laboratory strongly recommends micro-dissection of FFPE specimens to enrich for tumor cells. Many FFPE specimens contain only small amounts of tumor, which can significantly impact downstream results. We highly recommend you collaborate with a pathologist to circle the tumor area on the H&E. Visit our FAQ page to learn more about finding a pathologist.
Turnaround Time and Low Volume Requests
Isolations will be performed in the order they were received. The typical turnaround time is approximately 2 weeks after the Molecular Diagnostics laboratory has physically received the specimens, but may be as long as 4-6 weeks. If a request is submitted for fewer than 4 specimens, we will wait up to one month for additional samples to be requested. Alternatively, the investigator can pay for 4 samples, and the extraction will be processed in the order it was received.
Contact Us
BMP Shared Resource Director
Chris Fillmore, PhD
Biorepository Director
Chris Fillmore, PhD
chris.fillmore@hci.utah.edu
801-213-5787
Molecular Diagnostics Director
Nieca Bronson, MS, MLS, ASCP(CM)
nieca.bronson@hci.utah.edu
801-587-4406
Research Histology Director
Meisha Loveless, HT (ASCP) CM
meisha.r.loveless@aruplab.com
801-213-6088
Governance
HCI Senior Director Oversight
Alana Welm, PhD
Medical Director
Eric Snyder, MD, PhD
Faculty Advisory Committee Members
Dan Albertson, MD
Shannon Buckley, PhD
Nicola Camp, PhD
Howard Colman, MD, PhD
Randy Jensen, MD, PhD
Manish Kohli, MD
Charles Murtaugh, PhD