
Hematology Drug Discovery Program
We screen small molecules—often in development for other oncology indications—for their potential activity against acute myeloid leukemia (AML). After passing Phase I safety evaluation, molecules are screened using AML cell lines with and without stromal support. Promising agents are then tested in primary patient samples and xenograft models with SPECT/CT or PET/MRI imaging. Molecules showing in vivo activity may proceed to investigator-initiated clinical trials at Huntsman Cancer Institute.
All discussions and results are confidential and subject to CDA/NDA agreements. Contact Tony Pomicter to collaborate.

Tony Pomicter, MS
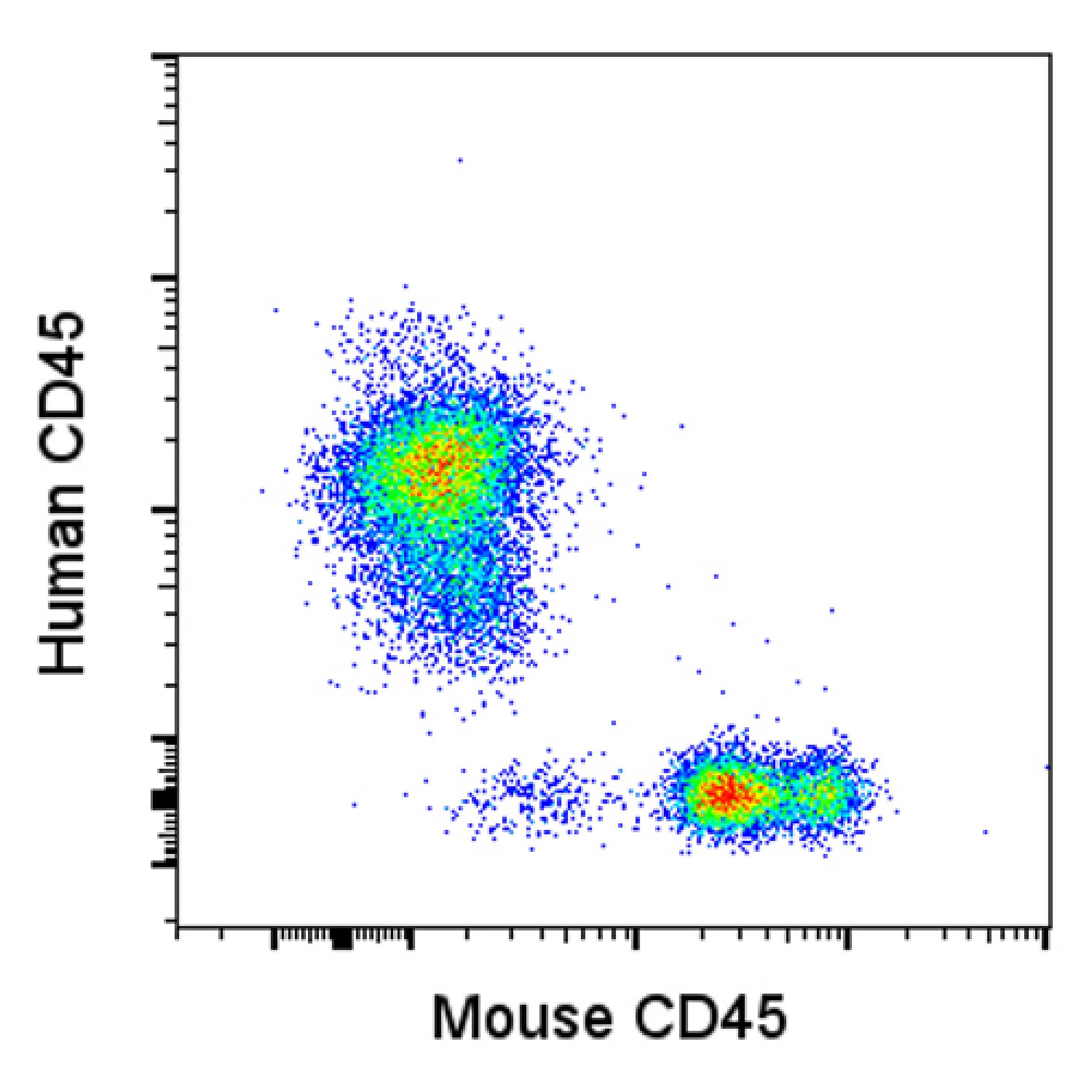
Flow cytometry analysis demonstrates engraftment of primary AML blasts into the bone marrow of a mouse.
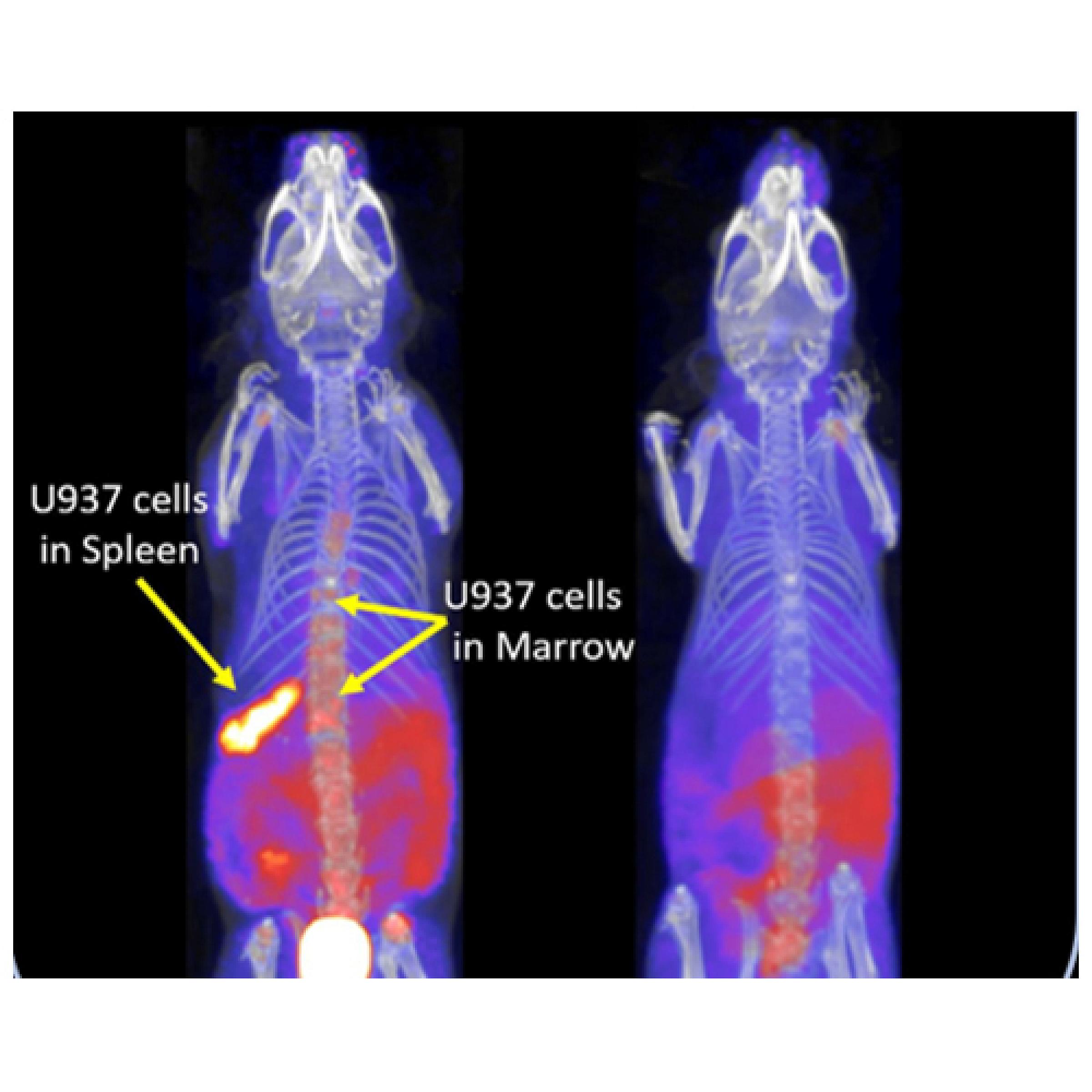
18F-fluorothymidine PET imaging with CT and MRI demonstrates leukemia engraftment in the vertebrae (left) of a mouse, which is resolved after 14 days of treatment (right).
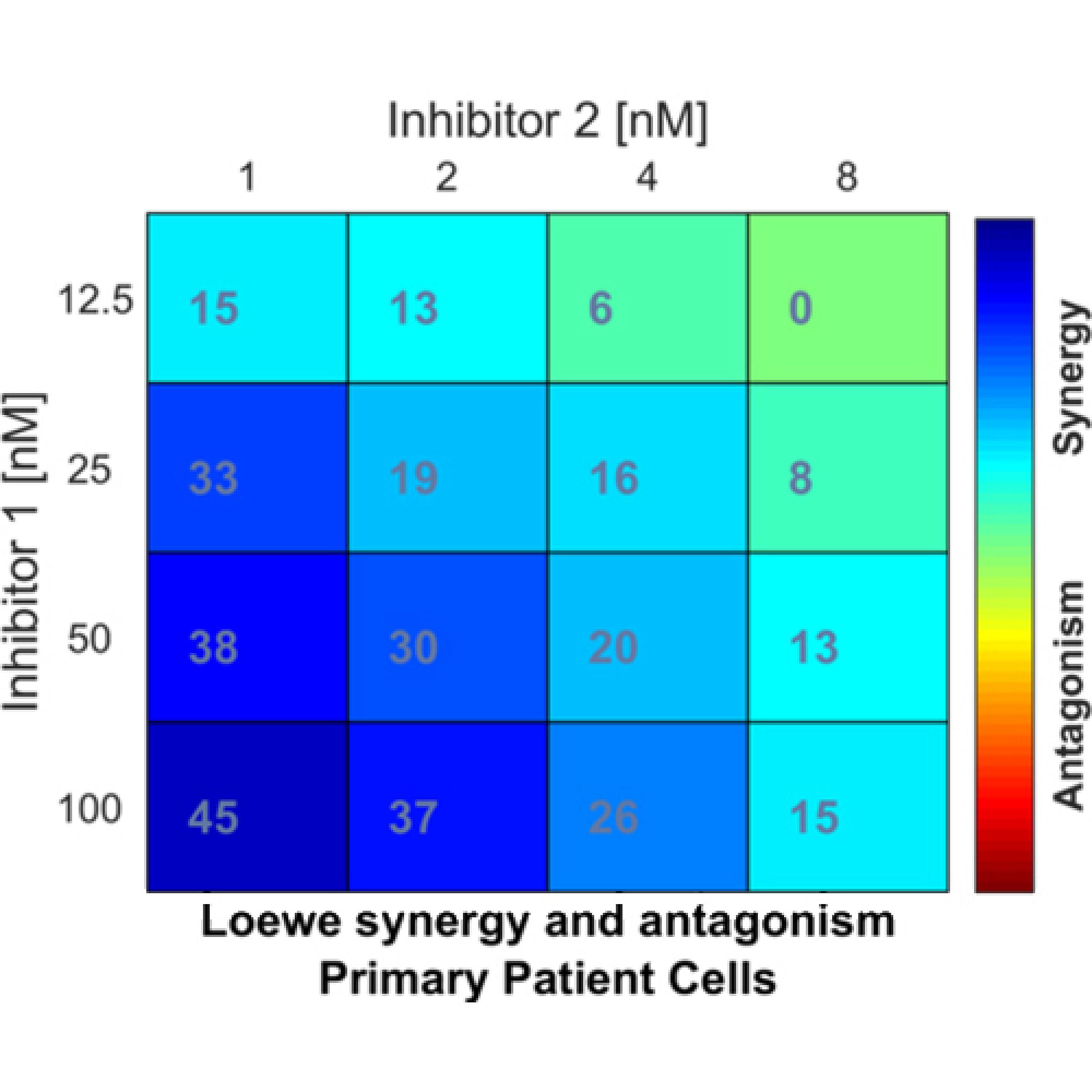
Inhibitor combination analysis in primary human CD34+ myelofibrosis cells demonstrates strong synergy (darker blue) at higher doses of Inhibitor 1 and lower doses of Inhibitor 2.
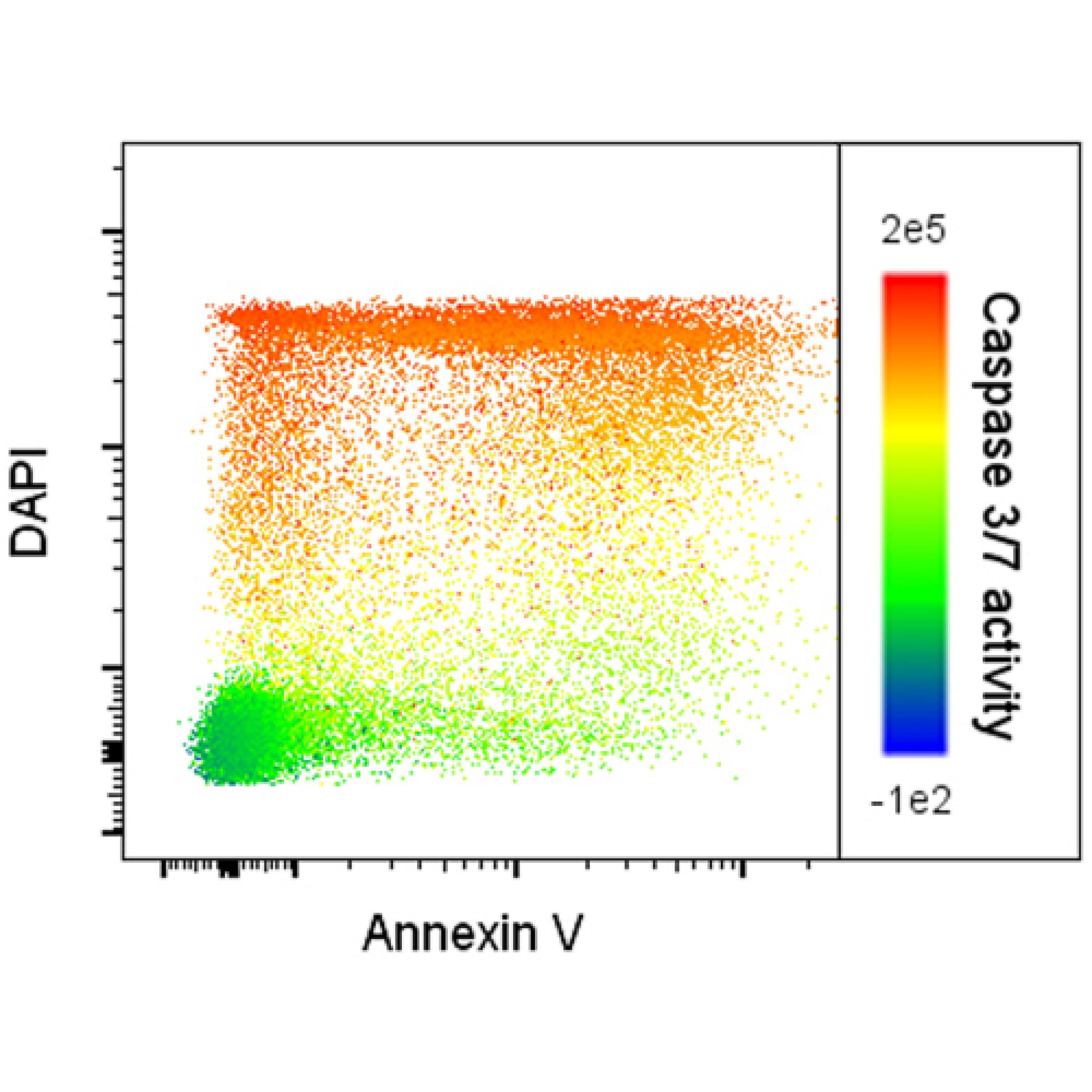
Apoptosis flow cytometry analysis of primary AML blasts demonstrates increasing activity of caspase 3 & 7 (color spectrum) with increasing DAPI-positivity (y-axis) but not Annexin V-positivity (x-axis).